Made by  Italy, posted in Chemistry, on October 19, 2012 1:10 am
Italy, posted in Chemistry, on October 19, 2012 1:10 am

LEARNING OBJECTIVES
? to define a chemical change
? to recognize a chemical change and distinguish it from a physical one
? to read a chemical equation
? to write chemical equations
? to balance chemical equations
? to classify chemical reactions
? to write formation and combustion reactions for given compounds.
? to recognize the limitant reactant
? to make stoichiometric calculations:
from mass to moles, from moles to mass,
yield, masses needed for complete reactions (from a given quantity of a reactant)
|
This material rate is 6 of 5 points from 1 votes.
|

 (26)
(26)
|
Made by  Turkey, posted in Chemistry, on March 19, 2013 2:37 am
Turkey, posted in Chemistry, on March 19, 2013 2:37 am

The software related w?th the Chem?stry formulas ?s used to re?nforce the top?c. The formulas related w?th the chem?stry are matched w?th the?r names.
|
This material rate is 0.2 of 5 points from 5 votes.
|

 (41)
(41)
|
Made by  Romania, posted in Chemistry, on August 14, 2012 2:21 am
Romania, posted in Chemistry, on August 14, 2012 2:21 am

Some Information about luminol
IUPAC name: 5-amino-2,3-dihydro-1,4-phtalazinedione
Molar mass:
177,16 g/mol
Melting point:
319 � 320 �C
|
This material rate is 0 of 5 points from 0 votes.
|

 (68)
(68)
|
Made by  Turkey, posted in Chemistry, on May 13, 2012 11:15 pm
Turkey, posted in Chemistry, on May 13, 2012 11:15 pm

Lesson Objectives
1. To be able to write the name of the ionic compound the formula of which is given.
2. To be able to write formula of the ionic compound the name of which is given.
3. To be able to write the name of the covalent compound the formula of which is given.
4. To be able to write the formula of the covanlent compound the name of which is given.
5. To use ICT as a successful tool when learning.
|
This material rate is 0.1 of 5 points from 7 votes.
|

 (124)
(124)
|
Made by  Turkey, posted in Chemistry, on May 12, 2013 10:50 pm
Turkey, posted in Chemistry, on May 12, 2013 10:50 pm

Procedures:
1.Students are given the definitions of Group and Period s.
2.They are taught systematic variying features at the Periodic Table.
3.Groups of A and B and their features are told.
|
This material rate is 0 of 5 points from 0 votes.
|

 (126)
(126)
|
Made by  Italy, posted in Chemistry, on May 12, 2013 9:50 pm
Italy, posted in Chemistry, on May 12, 2013 9:50 pm
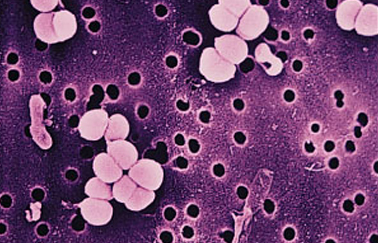
Objective of the activity is to determine the quality of water samples of different sources using chemical and microbiological indirect methods.
The action has planned activities in the laboratory of Chemistry with the determination of the presence of the organic substance, as an indicator of contamination, through the method of Kubel and in the laboratory of Microbiology with the determination of Total Coliforms with MPN method.
|
This material rate is 0 of 5 points from 0 votes.
|

 (155)
(155)
|

 Italy, posted in Chemistry, on October 19, 2012 1:10 am
Italy, posted in Chemistry, on October 19, 2012 1:10 am 

 Turkey, posted in Chemistry, on March 19, 2013 2:37 am
Turkey, posted in Chemistry, on March 19, 2013 2:37 am 
 Romania, posted in Chemistry, on August 14, 2012 2:21 am
Romania, posted in Chemistry, on August 14, 2012 2:21 am 


